Peritoneal Cancer
Peritoneal cancer, also known as peritoneal metastasis, refers to the spread of cancer to the peritoneum—a thin, protective membrane that lines the abdominal cavity and envelops the organs within it.


Peritoneal cancer, also known as peritoneal metastasis, refers to the spread of cancer to the peritoneum—a thin, protective membrane that lines the abdominal cavity and envelops the organs within it.


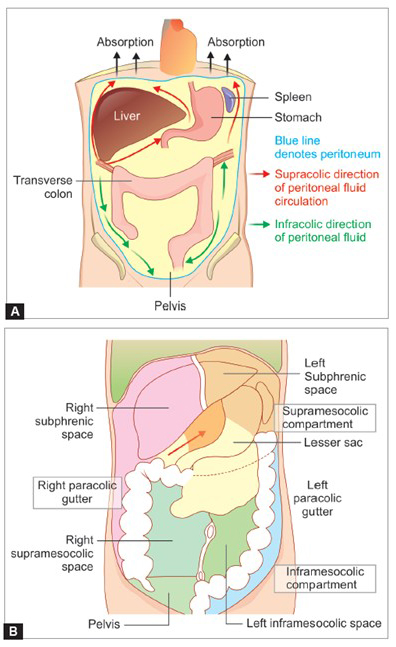
The peritoneum is a delicate membrane that forms a lining on the inner wall of the abdomen (parietal peritoneum) and also covers the abdominal organs (visceral peritoneum). The space between these two layers, known as the peritoneal cavity, contains a small amount of clear fluid that allows the internal organs, especially the intestines, to move freely and without friction.
Remarkably, the surface area of the peritoneum is equivalent to that of the skin—approximately 2 square meters.
The abdominal cavity is the largest hollow space in the body and houses most of the digestive organs including the stomach, intestines, liver, pancreas, spleen, kidneys, and adrenal glands. The diaphragm, a muscular partition, forms its upper boundary, separating it from the chest cavity, while its lower boundary merges into the pelvic cavity, which contains organs like the bladder, prostate, uterus, vagina, and rectum. Vertically, it is enclosed by the spine and abdominal musculature.
Peritoneal cancer refers to the presence of cancerous growths on the peritoneum. These tumor nodules can range in size—from tiny, grain-like deposits to larger masses over 5 cm. In most types of cancer, involvement of the peritoneum signifies stage 4 disease. However, in ovarian cancer, peritoneal involvement can present as early as stage 2 or 3.
More critical than the cancer stage is the extent of disease spread within the peritoneum. Much like the skin covers the body externally, the peritoneum covers a vast internal surface.
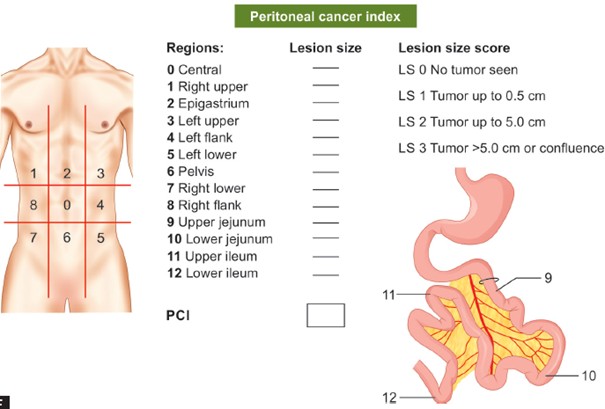
To quantify the disease, the peritoneum is divided into 13 anatomical regions. Each region is scored from 0 to 3 based on tumor size (0 = no tumor, 3 = tumor >5 cm). The sum of these scores gives the Peritoneal Cancer Index (PCI), which ranges from 0 to 39. A lower PCI is associated with improved outcomes following treatments like cytoreductive surgery and HIPEC (Hyperthermic Intraperitoneal Chemotherapy).
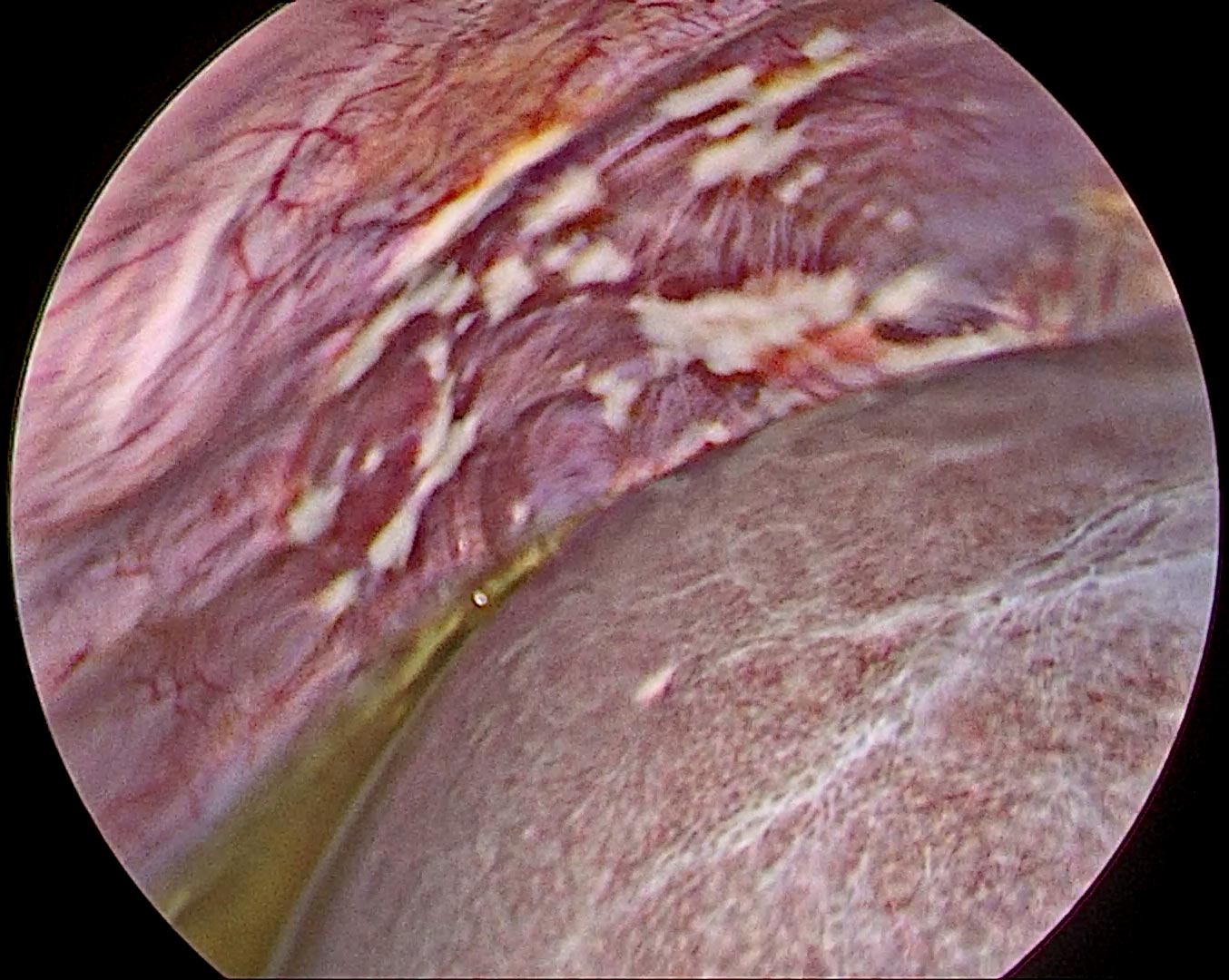
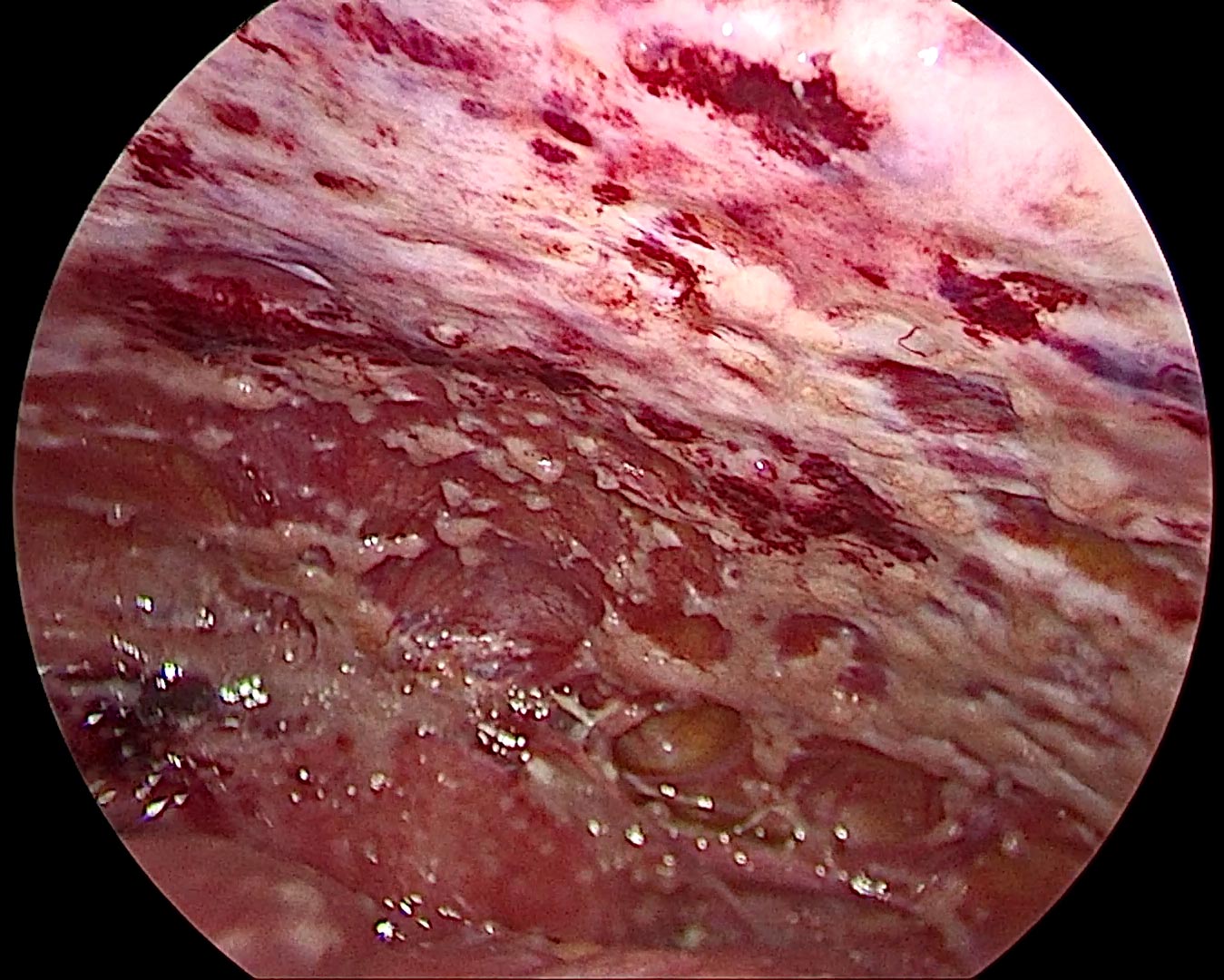
Peritoneal cancer can result in two major complications:
In healthy individuals, the peritoneum continuously produces and reabsorbs a small amount of lubricating fluid. This balance allows organs to glide smoothly against each other.
In peritoneal cancer, tumor deposits can interfere with this balance—often by obstructing absorption pathways—leading to a buildup of fluid known as ascites. This causes abdominal swelling, rapid weight gain, discomfort, breathing difficulties, fatigue, and loss of appetite.
Cancer nodules can form adhesions between loops of bowel and the abdominal wall, impairing bowel mobility. In some cases, tumors compress the intestines from the outside or block them entirely, disrupting the normal passage of food and gas. This results in symptoms such as cramping, bloating, vomiting, and constipation—collectively known as malignant bowel obstruction (MBO).
In contrast, a single tumor within the colon or rectum can also cause obstruction by narrowing the intestinal lumen, typically treated with surgery or stenting.
Traditionally, the treatment of peritoneal cancer relied on systemic (intravenous) chemotherapy, aimed at slowing disease progression and providing symptom relief. In cases where the cancer caused a blockage in the intestine, palliative surgery—such as creating a bypass around the obstructed bowel segment—was often performed to restore digestive function.
For patients with ascites (fluid buildup in the abdomen), medications were prescribed to reduce fluid production and enhance its removal. Symptomatic relief was provided by paracentesis (tapping), a procedure where fluid is drained from the abdominal cavity. However, with these conventional approaches, patients typically survived between 3 to 18 months and often experienced a significantly compromised quality of life.
Over the past 25 years, a new and more effective treatment approach has revolutionized the management of peritoneal carcinomatosis—Cytoreductive Surgery (CRS) combined with Hyperthermic Intraperitoneal Chemotherapy (HIPEC).
This method involves two key steps:
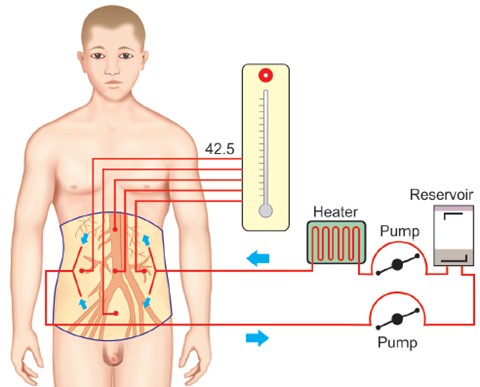
It’s important to understand that not all stage 4 cancers are the same. Patients with cancer confined to a single site—such as only the liver (in colorectal cancer) or only the peritoneum (in ovarian cancer)—often have a better prognosis compared to those with multiple organ involvement.
For patients whose cancer spread is limited to the peritoneum, CRS and HIPEC offer a potentially life-extending, and in select cases, curative option. Some patients undergoing this combined approach can enjoy long-term survival and return to a normal lifestyle.
The feasibility and outcome of treatment depend on several factors:
Peritoneal Cancer Index (PCI): The volume and distribution of tumor deposits within the peritoneum are critical. Patients with a low PCI and limited extra-peritoneal disease (e.g., 2–3 deposits in the liver or lungs) may still be candidates for surgery. In contrast, widespread disease may render CRS and HIPEC unfeasible.
Misdiagnoses, apprehensions about extensive surgery and the cost involved are the major barriers for a patient to contact a specialist on time. Awareness about peritoneal cancers and its treatment is thus important to enable people to seek the right treatment at the right time.
Dr. Shouptik Basu is actively involved in the treatment of peritoneal surface malignancies, with specialized expertise in Cytoreductive Surgery and HIPEC. Having trained at high-volume tertiary cancer centres in India and abroad, he brings a patient-centric and evidence-based approach to managing these complex cases.
He has successfully performed numerous CRS and HIPEC procedures and continues to evaluate and treat eligible patients with peritoneal metastases from various cancers including colorectal, appendiceal, and ovarian origins.
Through a combination of surgical skill, academic excellence, and compassionate care, Dr. Basu strives to offer selected patients with peritoneal cancer a genuine chance at extended survival—and, in some cases, long-term remission.
Ovarian cancer is the most frequent cause of peritoneal cancer among all cancers.
Ovarian cancer arises from the ovaries, which are a part of the female reproductive system. It is the fourth most common cancer in women and ranks eighth among all cancers in females. Most ovarian cancers begin from the surface lining of the ovaries, although recent studies indicate that a significant number may actually originate from the fallopian tubes, which are lined by similar cells.
In rare cases, it may begin from the peritoneum, the thin membrane that lines the abdominal and pelvic cavities.
Among all cancers, ovarian cancer has the highest tendency to spread to the peritoneum. Unlike other cancers where peritoneal spread is classified as stage IV, in ovarian cancer it is seen even in stage III.
This cancer is often detected late—about 80% of cases are diagnosed at an advanced stage—as it may grow silently without producing symptoms. The disease often becomes apparent only when it causes abdominal swelling, breathlessness, or difficulty in eating. Occasionally, it is detected early during scans performed for vague symptoms like abdominal pain or urinary issues.
The spread to the peritoneum can also happen very quickly, within just a few months.

The usual treatment involves both surgery and chemotherapy. Surgery includes removal of the uterus, ovaries, omentum, and other visible tumor-bearing areas. This is followed by six cycles of chemotherapy. This is called Surgical Staging.
In cases where the disease is too extensive for initial surgery, chemotherapy is given first, and surgery is done after 3–4 cycles. The remaining chemotherapy is completed post-surgery. This is called Interval Cytoreduction and HIPEC.
However, even with initial good response, 60–70% of patients relapse, often within the peritoneum. To reduce or delay recurrence, maintenance therapy for 1–2 years may be offered to select patients.
Regardless of whether chemotherapy or surgery comes first, radical surgery is essential for improving survival and delaying recurrence. Still, less than half the patients live beyond five years, as recurrence remains common.
The surgery typically performed in ovarian cancer is called debulking surgery. It removes the uterus, ovaries, omentum, and visible disease in a limited fashion. However, peritoneal disease may not be fully addressed, and often only small areas are removed.
Cytoreductive surgery, on the other hand, involves the removal of all peritoneal regions with tumor, and also areas that may harbor microscopic disease, such as the greater and lesser omentum. Even normal-appearing peritoneum is removed because of the high chance of hidden cancer cells. This improves chances of survival and reduces recurrence risk.

HIPEC (Hyperthermic Intraperitoneal Chemotherapy) is used along with radical surgery to treat microscopic disease that cannot be seen or removed surgically. It helps delay or prevent recurrence. Some studies show that HIPEC reduces peritoneal recurrence in ovarian cancer.
The most common drug used for HIPEC is Cisplatin. Since Cisplatin resistance is a major concern in ovarian cancer, the heat used during HIPEC may help overcome this resistance.
However, HIPEC is not suitable for:
Currently, HIPEC is recommended for select patients, especially those undergoing surgery after a few chemotherapy cycles (interval cytoreduction). In such patients, HIPEC may prolong life by a year or more.
Dr. Shouptik Basu is actively involved in the surgical management of ovarian cancer, including cytoreductive surgery and HIPEC. With specialized training in peritoneal surface malignancies, he carefully selects patients who may benefit from this aggressive treatment. Dr. Basu’s work in this area is aimed at improving survival and enhancing the quality of life in patients with advanced ovarian cancer. He has also done extensive research in this clinical domain and has developed AI based treatment algorithms for managing these patients
PIPAC: A Novel Approach in the Treatment of Peritoneal Cancer
Pressurized Intraperitoneal Aerosol Chemotherapy (PIPAC) is an innovative, minimally invasive technique used to treat patients with peritoneal carcinomatosis—a condition where cancer spreads to the lining of the abdominal cavity.
Unlike traditional chemotherapy, which is administered intravenously, PIPAC delivers chemotherapy directly into the peritoneal cavity in the form of a fine aerosol (mist) during a laparoscopic (keyhole) surgery. This allows the drug to cover the entire surface of the peritoneum more uniformly and in higher local concentrations, while minimizing side effects on the rest of the body.
The procedure is typically repeated every 6–8 weeks and is usually well-tolerated, even by patients who are not candidates for major surgery. It is especially useful in patients with recurrent or inoperable peritoneal disease from cancers such as ovarian, colorectal, gastric, and appendiceal cancer.
Dr. Shouptik Basu is trained in PIPAC and incorporates it into the treatment of select patients with peritoneal surface malignancies. This approach offers hope for improved quality of life and disease control in patients with limited treatment options.

NIPEC-LT is a treatment in which chemotherapy is delivered directly into the abdomen (peritoneal cavity) at normal body temperature, over multiple sessions. It is used for cancers that have spread to the peritoneum, such as colorectal, ovarian, or gastric cancers.
Peritoneal cancers are often resistant to regular (systemic) chemotherapy because the drugs in the bloodstream do not reach the abdominal cavity effectively. This is due to a natural barrier called the blood–peritoneal barrier. NIPEC-LT overcomes this by delivering chemotherapy directly where it’s needed.
Unlike HIPEC (Heated Intraperitoneal Chemotherapy), which is done once during surgery, NIPEC-LT is given repeatedly through a catheter placed in the abdomen, without the need for surgery each time.
This technique has gained support from recent clinical trials and is now advocated by Prof. Paul Sugarbaker, the pioneer of HIPEC. In fact, NIPEC (also called NIPS, in Japan) was used even before HIPEC was discovered by Prof. Sugarbaker, in Japan.
It was extensively practiced by Prof. Yonemura, a world leader in Gastric Cancer and Peritoneal Surface Oncology.
Dr. Shouptik Basu, who trained under Prof. Yonemura, now offers this treatment. He is a specialist in Peritoneal Surface Malignancies and performs NIPEC-LT as a cost-effective, evidence-based option for eligible patients.
Benefits:
Possible Side Effects: